|
Post an Offer to Sell New here? Please subscribe to post trade leads. It's FREE! |
Offers to Sell
Home > Offers to Sell
| Summary of 5/10/26 4:10 GMT: | >> Show Compact View |
Automatic Busbar Inspection Machine with High Voltage Withstanding Insulation Resistance Grounding Test Functions Busbar bending machines from us set the industry standard in precision and reliability. Whether you’re bending copper busbar, aluminum bar, steel bar, or any type of metal bar stock, our proprietary technology delivers the consistency and efficiency that today’s businesses demand. Overview The equipment is used for busbar online inspection. To improve the busbar production level and quality. To decrease the labor’s working force and improve the working efficiency and production efficiency. Busbar Inspection Machine Function introduction Busbar Automatic Production Line is composed of three parts, feeding positioning station, inspection station and discharge station. When the busbar enters the feeding position, the control system will select the right testing fixture to start testing from the information identified by bar code or previous assembly line. Three tests will be performed including HV withstanding test, insulation resistance test and grounding test. After test is finished, testing report will be generated. Busbar Inspection Machine Equipment data on automatic busbar inspection line No. Parameters Model KD-JCX/NC-E/H-6 Busbar Height 100mm~650mm Busbar Length 1200mm~6000mm Busbar Type Straight length type Position Type Unilateral / Middle Working Efficiency 2 min/piece(testing time is adjustable.) Servo Nos 2 axis( Z axis/X axis) Vision System Phase error judgment( optional) Printing System Bar code printing/ Report printing( optional) High Voltage Withstanding Test 0~5000V Insulation Test 1~1000V Grounding Test 0~25A Power 380~415VAC 50~60Hz 5kW Equipment Dimension L12000mm*W1200mm*H1900mm Automatic Inspection Straight Length Busbar Machine , Busbar Fabrication Machine 0 Automatic Inspection Straight Length Busbar Machine , Busbar Fabrication Machine 1
Phone:
Fax:
Email:
NO 123 Suzhou Dadao East Road Suzhou Jiangsu China
Suzhou 215000
China
Mini type 1xN 2xN PLC splitter with connectors TELCORDIA 1209&1201 STANDARD Dawnergy’s planar light wave circuit (PLC) splitter is optical power management device which uses silica optical waveguide technology. It has small size, high reliability, wide operating wavelength range. It is widely used in PON networks and realizes optical signal power splitting. Features: Low insertion loss Low PDL Good channel uniformity High-reliability and stability Qualified according to Telcordia 1209 and 1221 requirements Parameters 1x2 1x4 1x8 1x16 1x32 1x64 Operating Wavelength(nm) 1260~1650 Insertion Loss (dB) Typical 3.6 6.8 10 13.1 16.2 20 Max(P/S) 3.8/4.0 7.2/7.4 10.5/10.7 13.6/13.7 16.5/16.8 20.5/21.0 Uniformity (dB) Typical 0.3 0.4 0.5 0.6 0.8 1.2 Max 0.4 0.6 0.8 1.2 1.5 2.5 PDL (dB) Typical 0.1 0.1 0.15 0.15 0.15 0.2 Max 0.2 0.2 0.3 0.3 0.3 0.4 Return Loss (dB) Min 55 Directivity (dB) Min 55 Fiber Length (m) 1 (+/-0.05) or customer specified Fiber Type ITU-T G657A1 or customer specified WDL (dB) Typical 0.2 0.2 0.2 0.3 0.3 0.3 Max 0.3 0.3 0.3 0.5 0.5 0.5 TDL (dB) (-40~85℃) Typical 0.2 0.2 0.2 0.3 0.3 0.3 Max 0.4 0.4 0.4 0.5 0.5 0.5 Operating Temperature (℃) -40~85 Storage Temperature (℃) -40~85 Packaging Size(mm) Bare Splitter 40x4x4 50x7x4 60x12x4 Mini Module 60x7x4 60x12x4 80x20x6 100x40x6 Cassette Type 100x80x10 120x80x18 Parameters 2x2 2x4 2x8 2x16 2x32 2x64 Operating Wavelength(nm) 1260~1650 Insertion Loss (dB) Typical 3.7 7 10.8 13.6 16.8 20.5 Max(P/S) 3.9/4.2 7.5/7.8 11.2/11.5 14.2/14.5 17.4/17.7 21.0/21.5 Uniformity (dB) Typical 0.5 1 1 1.2 1.5 2.2 Max 0.6 1.2 1.5 1.8 2 2.7 PDL (dB) Typical 0.1 0.1 0.2 0.3 0.3 0.3 Max 0.2 0.2 0.4 0.4 0.4 0.4 Return Loss (dB) Min 55 Directivity (dB) Min 55 Fiber Length (m) 1 +/-0.05 or customer specified Fiber Type ITU-T G657A1 or customer specified WDL (dB) Typical 0.3 0.3 0.3 0.5 0.5 0.8 Max 0.4 0.4 0.5 0.6 0.8 1.2 TDL (dB) (-40~85℃) Typical 0.3 0.3 0.3 0.4 0.4 0.4 Max 0.4 0.4 0.4 0.5 0.5 0.5 Operating Temperature (℃) -40~85 Storage Temperature (℃) -40~85 Packaging Size(mm) Mini Module 60x7x4 80x12x5 100x20x6 100x40x6 Notes: 1. Measurements are done at room temperature. Connector loss is excluded in the above specifications. 2. S=Standard grade, P=Premium grade
Phone:
Fax:
Email:
No. 500 Songmin Road, Songjiang District, Shanghai,China.
Shanghai 200000
China
How to make an order : 1. Name what your need ( which product , quantity ) 2. Full payment would be done once we come to an agreement on all details . 3. Parcel would be shipped out upon receipt of payment . 4. Info about parcel would be offered ( Photo , tracking number ) 5. Finally you would get the parcel in 3~7 days . Our service: 1.We have experience in exporting Pharmaceutical intermediates . 2.Professional packing with professional materials 3. We have products in stock, and we will deliver them soon when your PO arrived
Minimum Order: 1 bags
Phone:
Fax:
Email:
Handan, Hebei
Handan 056000
China
independently developed by Beilu Pharma for the treatment of generalized anxiety disorder (GAD). It is a unique anti-GAD Chinese medicine approved by NMPA. The clinical efficacy of Jiuweizhenxin Granules in treating GAD is equivalent to that of Paroxetine tablets, but the adverse reactions of Jiuweizhenxin Granules are significantly lower than that of Paroxetine. Clinical research results show that taking Jiuweizhenxin Granules alone for 6 weeks can significantly improve the anxiety symptoms of patients and improve the quality of life with continuous use. Features Of anti anxiety traditional Chinese medicine-Jiuweizhenxin Granules Product name: Jiuweizhenxin Granules Ingredients: Ginseng(rhizome-removed), Wild jujube seed, Schisandra Chinensis, Poria cocos, polygala, rhizoma corydalis, Radix Asparagi, Cooked rehmannia root, cinnamon Specification: 6g/ bag Indication: Nourishing heart and tonifying spleen: the product is intended for generalized anxiety disorder, syndrome of deficiency of both heart and spleen, symptoms of unrelieved thoughtfulness and excessive worries, insomnia, dreaminess and palpitations. Benefiting vital energy and soothing the nerves: small and wiry pulse or deep thready pulse, sallow complexion, pale tongue with thin white coating, dizziness, sweating, frequent sighs, loss of appetite, fatigue weariness. Dosage and Administration: Administered with warm water, 1 bag each time, three times a day. Adverse Reactions: Occasionally dry mouth, hazy vision, increased constipation, nausea and vomiting, diarrhoea, inappetence or anorexia, abdominal distension, bitter mouth, gastric pain, somnolence, agrypnia, tremor, headache, wooziness, lipothymia, electrocardiogram abnormal, palpitations, tachycardia, ALT increased, white blood cells decreased, and menstrual disorders. Shelf life: 36 months The Effect of Jiuweizhenxin Granules Jiuweizhenxin Granules is a modern Chinese patent medicine approved by NMPA to treat anxiety disorders. The ingredients of Jiuweizhenxin Granules are Ginseng(rhigome-removed), Wild jujube seed, Schisandra Chinensis, Poria cocos, polygala, rhizoma corydalis, Radix Asparagi, Cooked Rehmannia root, cinnamon. The anti-anxiety effect is definite, especially for physical anxiety such as diarrhoea, headache, dizziness, and insomnia. It has a unique overall regulating effect. As one of the effective anti-anxiety drugs on the market, Jiuweizhenxin Granules can significantly improve anxiety and depression symptoms. If combined medication can significantly improve depression symptoms compared with the single medication group, the combination of Jiuweizhenxin Granules and duloxetine can significantly improve the clinical efficacy. Not only that, Jiuweizhenxin Granules can significantly improve the symptoms of insomnia in patients with insomnia. After taking Jiuweizhenxin Granules for 4 weeks, the time to fall asleep, sleep time, sleep quality, daytime dysfunction and total sleep score is better than the treatment before 【Note】Use with caution in patients with abnormal cardiac and liver functions and leukopenia.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
Repaglinide tablets are non-sulfonylurea insulin secretion agents and are used to treat patients with type 2 diabetes. Repaglinide tablets were listed in China in 2000, and it is a Class B drug in the National Medical Insurance List. Repaglinide tablets have been listed as a drug for diabetic patients with renal insufficiency by many guidelines at home and abroad. Product name: Repaglinide Tablets Characteristic: White or off-white tablet Specification: repaglinide 0.5 tablet×30 tablets, repaglinide 0.5mg×60 tablets. Indications: It is used for type 2 diabetes (non-insulin-dependent) patients whose hyperglycemia cannot be effectively controlled by diet control, weight loss and exercise. When metformin alone cannot effectively control hyperglycemia, Repaglinide tablets can be combined with metformin. Treatment should start from the adjuvant treatment of diet control and exercise to reduce blood sugar during meals. Specifications of Repaglinide Tablets: 0.5mg Usage and Dosage,Adverse reactions,Contraindications,Precautions: , Please refer to the package insert. Medicine Interactions: Some drugs are known to affect glucose metabolism. Therefore, doctors should consider possible drug interactions. In vitro studies have shown that the metabolism of Repaglinide is affected by CYP2C8 and CYP3A4. Repaglinide was found to be a substrate for active liver uptake (organic anion transporter OATP1B1). Data from clinical studies carried out in healthy volunteers show that CYP2C8 is the main enzyme in the metabolism of Repaglinide, while CYP3A4 has limited effects. But if the effect of CYP2C8 is inhibited, the effect of CYP3A4 will be relatively enhanced. Therefore, the metabolism and clearance of Repaglinide may be changed due to the inhibition or induction of cytochrome P450. Therefore, extreme caution should be exercised when using CYP2C8 and CYP3A4 inhibitors simultaneously with Repaglinide. Drugs that inhibit OATP1B1 (such as cyclosporine) may increase the plasma concentration of Repaglinide. ( For more details, please refer to the package insert.) Storage: Similar to the storage of glimepiride tablets, tab repaglinide should also be shaded, sealed, kept in a dry place. Packaging: Aluminum plastic packaging, 15 pills/plate , 2 plates/box; 15 pills/plate, 4 plates/box, 15 pills/plate, 6 plates/box Beilu is a professional contrast media manufacturer, we provide repaglinide 0.5 mg tablet, ct contrast media and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
Iohexol Injection is a non-ionic Contrast Media. Compared with a traditional ionic Contrast Media, Iohexol Injection has an excellent molecular structure and it can greatly improve user safety and significantly reduce adverse reactions. The iodinated contrast agent has excellent clinical feedback and good imaging results. In 1998, Iohexol Injection was launched as Beilu's 1st iodinated Contrast Media. Product name: Iohexol Injection Generic name: Iohexol Injection Type: Injectable Contrast Media for CT/X-ray Ingredients: Iohexol Specification: (1) 20 ml: 6 gI (2) 50 ml: 15 gI (3) 75 ml: 22.5 gI (4) 100 ml: 30 gI (5) 20 ml: 7 gI (6) 50 ml: 17.5 gI (7) 100 ml: 35 gI Administration: For injection or oral use of iodinated contrast Adverse reactions: See the details from the package insert Shelf life: 36 months Indications of Iohexol Injection The omnipaque iohexol is an X-ray contrast agent. It can be used for angiocardiography, arteriography, urography, enhanced CT examination inclduing cervical, thoracic and lumbar myelography, CT cisternography following intrathecal injection (i.e., an injection into the subarachnoid space); arthrography, endoscopic retrograde cholangiopancreatography (ERCP), herniography or fistulography, hysterosalpingography, sialography, percutaneous transhepatic cholangiography (PTC), sinography, gastroenterography, T-tube angiography, etc. Precautions of Iohexol Injection General precautions for the use of nonionic-monomer contrast agent: 1. Special attention should be paid to patients with allergies, asthma and adverse reactions to iodinated contrast agents. Prophylactic medications such as steroids and H1/H2 histamine receptor antagonists may be considered for these patients. 2. The risk of serious reactions related to this type of CT contrast agent is low. However, iodinated contrast agents can provoke anaphylactoid reactions or other allergic reactions. Therefore, first aid measures should be trained in advance, and necessary rescue drugs and devices should be prepared for possible serious reactions. 3. In view of the low accuracy of pre-test in predicting allergic reactions caused by nonionic contrast agents and the possibility that the pre-test itself may lead to serious allergic reactions, it is not recommended to use pretest to predict iodine allergic reactions. 4. Venous access should be kept smooth throughout the X-ray examination. 5. In the in-vitro tests, the effect of nonionic contrast agents on the coagulation system is lighter than that of ionic contrast agents. During angiography, great care should be taken in the intravascular technical procedures, and the catheter should be irrigated with heparinized saline from time to time to reduce the thrombosis and embolism related to the technical procedures. 6. Patients, especially patients with multiple myeloma, diabetes, renal insufficiency, infants and young children, and the elderly, must be appropriately hydrated before and after the use of contrast agent. Electrolyte disturbance and hemodynamic disorders are common in infants (< 1 year old), particularly neonates. Special attention should be paid to patients with serious heart diseases and pulmonary arterial hypertension because they are prone to developing hemodynamic disorders and cardiac rhythm disturbances. 7. Patients with acute encephalopathy, brain tumour or history of epilepsy should be prevented from seizures, and special attention should be paid to them. The risks of seizures and neurologic reactions are significantly increased in patients with alcohol or drug abuse. Transient hearing loss or deafness occurs in a few patients after myelography, which is possibly caused by a decrease in CSF pressure after lumbar puncture. 8. In view of the high risks of acute renal failure after the use of contrast agent, special attention should be paid to patients with pre-existing renal impairment and diabetes and patients with atypical globulinemia (multiple myelomatoses and Waldenstrom macroglobulinemia). 9. Preventive actions include: Identify patients with high-risk factors. Ensure patients are appropriately hydrated and, if necessary, the infusion may be maintained intravenously prior to examination until the contrast agent is cleared from the kidneys. Avoid any nephrotoxic agent, oral gallbladder contrast agent, surgery for arterial occlusion, renal angioplasty or other major surgeries that increase the renal burden until the contrast agent is cleared. Repeat radiographic testing is delayed until renal function returned to the pre-examination level. To prevent lactic acidosis, the level of serum creatinine must be determined before intravascular injection of iodinated contrast agent into diabetic patients taking metformin. For patients with abnormal serum creatinine/renal function, metformin must be discontinued and the examinations with contrast agent must be postponed until after 48 hours of discontinuation. Metformin should be reused only after renal function and serum creatinine level are constant. In some emergency cases of patients with abnormal or unknown renal function, the physician must assess both advantages and disadvantages of using contrast agent and take precautions, such as discontinuation of metformin, adequate hydration of patients, testing of renal functions, and careful observation of symptoms of lactic acidosis. There is a potential risk of transient hepatic dysfunction. Special attention should be paid to patients with serious hepatic and renal insufficiency due to significantly longer time to clear contrast agent. Hemodialysis patients may receive the examinations with contrast agents. Immediate hemodialysis is not necessary after the injection of contrast agent because there is no evidence showing that hemodialysis can protect patients with renal impairment from contrast-induced nephropathy. Iodinated contrast agents may exacerbate symptoms of myasthenia gravis. Patients with pheochromocytoma should be treated with α-blockers to prevent hypertensive crisis in interventional procedures. Special attention should also be paid to patients with hyperthyroidism. Patients with multinodular goitre may develop hyperthyroidism after using iodinated contrast agents. The possibility of transient hypothyroidism in preterm infants after using contrast agents should be clearly recognized. Contrast media extravasation (CME) occasionally causes localized pain or oedema, which gradually subsides without sequelae. However, inflammation and even tissue necrosis may occasionally occur. Routine treatment is to elevate the affected limb and cold-compress at local sites. Surgical decompression is required in case of compartment syndrome. This type of Iohexol contrast agent should be drawn into the barrel before use. Each vial is for single use only, and the unused portion should be discarded. Beilu is a professional contrast media manufacturing companies, we provide ct contrast medium, iohexol package insert, omnipaque package insert and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
Glimepiride Tablets is a sulfonylurea hypoglycemic drug that can be used in combination with insulin. In 2001, Glimepiride tablets (Dibei), as the first hypoglycemic product of Beilu Pharma, was first listed in China and belongs to the second class of national new drugs. Product name: Glimepiride Tablets Specifications: glimepiride 1 mg×30 tablets;glimepiride 2 mg×15 tablets; glimepiride 2mg×30 tablets Characteristics: White tablet Indications : As one of the oral antidiabetic drugs produced by Beilu Pharma, our glimepiride tablets are only suitable for type 2 diabetes that cannot fully control blood sugar through diet control, exercise therapy and weight loss. Glimepiride tablets are not suitable for type 1 diabetes (for example, the treatment of diabetic patients with a history of ketoacidosis), diabetic ketoacidosis or Treatment of pre-diabetic coma or coma. Usage and Dosage: Please refer to the package insert Adverse reactions, Contraindications, Precautions: Please refer to the package insert. Storage: Shade, seal, and keep in a dry place. Packaging: Aluminum plastic packaging, 15 pills/plate, 1 plate/box; 15 pills/plate, 2 plates/box. Shelf life: 24 months Advantages of Glimepiride Tablets Double effect directly hit the hypoglycemic core Double effect: Glimepiride tablets can promote insulin secretion and improve insulin resistance. At the same time, it can improve the two major pathophysiological abnormalities of insulin secretion and insulin resistance in patients with type 2 diabetes. Efficiently reduce postprandial blood glucose, fasting blood glucose and glycosylated haemoglobin Glimepiride tablets monotherapy can comprehensively reduce FPG PPG and HbA1c in newly diagnosed T2DM patients Combination therapy, such as combined metformin, acarbose, etc., has a more significant overall hypoglycemic effect Less hypoglycemia, less weight impact, high safety Short-term treatment, the risk of glimepiride hypoglycemia is only 1/3 of that of Glibenclamide Long-term treatment, the risk of glimepiride hypoglycemia is only 1/7of that of Glibenclamide Glimepiride has a small impact on the weight of T2DM patients Once a day to improve patient compliance If the patient's compliance is poor, Glimepiride tablets should be selected, once a day. Beilu is a professional contrast media manufacturing companies, we provide gadolinium contrast media, glimepiride 2mg tablets, glimepiride package insert and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
Ferric Ammonium Citrate Effervescent Granules was launched in 2004. It is an oral magnetic resonance contrast agent/media approved by NMPA in the market, which is a safe and effective double contrast media product with the advantages of convenient use and good safety. This ammonium iron citrate can significantly improve the images quality of MRCP and MRU, and can also be used as a positive contrast agent for the digestive tract. Product name: Ferric Ammonium Citrate Effervescent Granules Type: Oral MRI Contrast Media Ingredients: Ferric ammonium citrate Specification: 3g: 0.6g(Fe 129mg), 6g: 1.2g(Fe 258mg) Posology and method of administration: Oral Adverse reactions: Foreign clinical data reported that after using this MRI contrast medium, 0.1-0.5% of patients has diarrhea, 0.1% had nausea, vomiting, and decreased appetite, and a very small number of patients has stomach discomfort and bloating. All the above symptoms can be eliminated by themselves. Shelf life: 24 months Indications of Ferric Ammonium Citrate Effervescent Granules Used for magnetic resonance abdominal imaging, angiography of the digestive tract (stomach duodenum and jejunum). Precautions of Ferric Ammonium Citrate Effervescent Granules Use with caution in the following patients: patients with the gastrointestinal tract such as peptic ulcer, ulcerative colitis, local enteritis, etc. The stool appearing black after administration is a normal phenomenon. During the ferric ammonium citrate uses, false-positive for occult blood may occur. Beijing beilu pharmaceutical co ltd is a professional contrast media manufacturing companies, we provide contrast medium, mri contrast medium and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
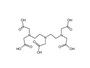
CAS No.: 67-43-6 Molecular formula: C₁₄H₂₃N₃O₁₀ Molecular weight: 393.35 Quality standard: CP Characteristics: White powder Content (HPLC): ≥99.0% Packing: 2.5KG/ drum Storage: Protected from light, sealed tightly Beijing beilu pharmaceutical co ltd is a professional contrast media manufacturing companies, we provide ct contrast agent, mri contrast medium, dtpa diethylenetriaminepentaacetic acid and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
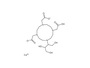
CAS No.: 151878-23-8 Molecular formula: C18H32CaN4O9 Molecular weight: 488.5461 Quality standard: CP Characteristics: White or off-white powder Content (HPLC): ≥99.0% Packing: 2KG/ drum Storage: Protected from light, sealed tightly Beilu is a professional contrast media manufacturer, we provide ct contrast agent, mri contrast medium and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
Product Description Brand New Fully Automatic Package Bobbin Dyeing Machine QR241D series is suitable for dyeing and pre-treatment of polyester and blended package yarn under high temperature conditions.It is also suitable for dyeing and pre-treatment of package yarn of cotton, hemp, acrylic, wool and other fibers under normal temperature conditions.Replace different dyeing carrier,It can be used for dyeing and pre-treatment various forms of nylon yarn,hank yarn,all types of tape cords,loose fiber and so on. Technical parameters and details: Design pressure:0.44Mpa Maximum working pressure:0.4Mpa Design temperature :140℃ Maximum working temperature :135℃ Heating rate:20℃~130℃ about 35 minutes(Saturated vapor pressure:0.7Mpa)Cooling rate:130℃~80℃ about 25 minutes(Cooling water pressure:0.3Mpa)Cooling water temperature:5℃~10℃ Liquid ratio:1:5-1:6 Model and specification: Item Model Main Tank diameter(mm) Total no. Of spindle Number Of Layers Maximum Capacity(KG) Motor power(KW) Lifting height (mm) 1 QR241D-40 400 3 4 12 4.0 2770 2 QR241D-43 430 4 4 16 5.5 2990 3 QR241D-43 430 4 6 24 5.5 3670 4 QR241D-50 500 5 5 25 5.5 3340 5 QR241D-50 500 5 6 30 7.5 3680 6 QR241D-50 500 5 7 35 7.5 4010 7 QR241D-53 530 6 6 36 7.5 4005 8 QR241D-53 530 6 9 54 11.0 5025 9 QR241D-65 650 8 9 72 15.0 4345 10 QR241D-75 750 12 7 84 11.0 4395 11 QR241D-75 750 12 9 108 15.0 5140 12 QR241D-85 850 18 7 126 15.0 4510 13 QR241D-85 850 18 9 162 18.5 5190 14 QR241D-90 900 19 7 133 18.5 4510 15 QR241D-90 900 19 9 171 18.5 5190 16 QR241D-90 900 19 12 228 22.0 6210 17 QR241D-100 1000 22 7 154 18.5 4560 18 QR241D-100 1000 22 9 198 22.0 5240 19 QR241D-100 1000 22 12 264 30.0 6260 20 QR241D-105 1050 24 9 216 22.0 6000 21 QR241D-110 1100 30 9 270 30.0 5670 22 QR241D-110 1100 30 10 300 30.0 6000 23 QR241D-110 1100 30 12 360 37.0 6680 24 QR241D-120 1200 36 9 324 37.0 5720 25 QR241D-120 1200 36 10 360 37.0 6050 26 QR241D-120 1200 36 12 432 45.0 6730 27 QR241D-140 1400 46 10 460 45.0 6140 28 QR241D-140 1400 46 12 552 45.0 6820 29 QR241D-140 1400 46 14 644 55.0 7500 30 QR241D-150 1500 56 10 560 55.0 6200 31 QR241D-150 1500 56 12 672 55.0 6880 32 QR241D-150 1500 56 14 784 75.0 7560 33 QR241D-170 1700 72 10 720 75.0 6660 34 QR241D-170 1700 72 12 864 75.0 7340 35 QR241D-170 1700 72 14 1008 90.0 8020 36 QR241D-186 1860 90 12 1080 90.0 7400 37 QR241D-186 1860 90 14 1260 110.0 8080 38 QR241D-186 1860 90 16 1440 110.0 8080 Remark: Standard package yarn dimension: 165mm(diameter) x 165mm(height) Spacing of spindle:170mm Weight of per package yarn: approach 1KG.
Phone:
Fax:
Email:
Room 2809-2810,Building B,Wanda Plaza,HuiShan District.Wuxi City,Jiangsu Province,China.
Wuxi 214000
China
Since Gadopentetate Dimeglumine Injection was launched in 1992 by Beilu Pharma in China, it was highly recommended and accepted by the radiologist, and quickly occupied the market with its excellent quality and reasonable price. Up to now, linear gadolinium has been safely used for nearly 30 years, with excellent enhancement effects and few side effects. Product name: Gadopentetate Dimeglumine Injection Generic name: Gadopentetate Dimeglumine Injection Type: Injectable Contrast Media for MRI, Linear Contrast Media No.1 market share in China Ingredients: Gadopentetate Dimeglumine Specification: 10ml:4.69 g; 12ml:5.63 g; 15ml:7.04 g; 20ml:9.38 g Posology and method of administration: Intravenous injection Adverse reactions: See the details from the package insert Shelf life: 36 months Indication of Gadopentetate Dimegumine Injection Magnetic resonance imaging of the central nervous system (brain and spinal cord), abdomen, chest, pelvis, limbs and other human organs and tissues. Precautions of Gadopentetate Dimegumine Injection 1. Use with caution in patients with severe kidney damage, epilepsy, hypotension, asthma and other allergic respiratory diseases and those with allergic tendencies. 2. Take care to avoid the extravasation of the gadopentetic acid liquid during injection to prevent tissue pain. 3. The serum iron and bilirubin levels of some patients will increase slightly after medication, but they are asymptomatic and can return to normal within 24 hours. 4. Pregnant women and breastfeeding women should use it with caution. Animal experiments show that a small amount of medicine liquid enters the milk. 5. The effective enhancement time of Magnevist contrast is 45 minutes. MRI examination should be performed immediately after intravenous injection. 6. The remaining medicine liquid after one examination should not be used again. 7. When applying this type of MRI contrast media, follow the relevant safety regulations in magnetic resonance imaging. 8. GBCAs should be used with caution. When plain scan MRI cannot obtain the corresponding vital diagnostic information, GBCAs can be used, and the lowest approved dose is used as much as possible. 9. Gadolinium deposition Current evidence shows that after repeated use of GBCAs, trace amounts of gadolinium can remain in the brain and other body tissues. Research reports have shown that multiple uses of GBCAs can increase the intensity of brain signals, especially in the dentate nucleus and globus pallidus. Currently, there are more reports about linear GBCAs and fewer reports about macrocyclic GBCAs. Animal experiments have shown that the amount of gadolinium deposited after repeated use of linear GBCAs is higher than that of repeated use of macrocyclic. The clinical significance of brain gadolinium deposition is unclear. In order to minimize the potential risks associated with gadolinium deposition in the brain, it must be used in strict accordance with the indications and approved doses. It is recommended to use the lowest approved dose that meets the requirement of diagnosis and perform a careful benefit-risk assessment and patient informed communication before repeated administration. Bei lu is a professional contrast media manufacturing companies, we provide gadopentetate dimeglumine, mri contrast medium, contrast media products and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
Model: GLD-FL101 Light Source: Integral LED Module Power: 10W/20W/30W/50W/100W/150W/200W/300W/400W Led Chips: SMD3030 14pcs/28pcs/36pcs/48pcs/72pcs/96pcs/108pcs/144pcs< , br>Luminaire Efficacy: 100-120lm/W CCT: 4000K (3000K/5000K/5700K upon request) CRI: > 70/ > 80 (upon request) Voltage: AC180-240V Lifetime: 100,000 hours at 25℃ ambient temperature Driver: Driverless
Minimum Order: 100
Phone:
Fax:
Email:
No.18,Meiwang Road,Ninghai Industrial Zone, Ningbo,China
Ningbo 315600
China
The main component of CT/X-ray Contrast Media is iodine. The Iodinated Contrast Media is a colorless, transparent, slightly viscous liquid. At present, the CT contrast agents can be mainly divided into ionic and non-ionic, according to the osmotic pressure can be divided into hypertonic, isotonic and hypotonic. Currently, non-ionic, isotonic or hypotonic Contrast Media are used clinically. When the CT contrast medium is injected, 97% of the contrast media products can be directly excreted from the kidneys within 1 day. Types of Contrast Media Iohexol Injection Iohexol Injection is non-ionic contrast agent. Compared with traditional ionic contrast agent, Iohexol Injection has the excellent molecular structure, it has greatly improved safety, and significantl... Iopamidol Injection Iopamidol Injection is the second-generation non-ionic iodinated contrast media launched in the global market. In 2015, Beilu firstly launched and listed this product in the Chinese market. Product name: Iopam... Iodixanol Injection Iodixanol Injection is a Contrast Media that is isotonic with plasma. It has excellent cardiac safety and renal safety, which is most comfortable to use, especially suitable for high risk patients. Reasonable Use of Iodinated Contrast Agents Evaluate patient benefits/risks before use History of allergies: Patients who have previously experienced moderate to severe anaphylactoid reactions to iodinated contrast agents or patients with a history of allergies requiring treatment are at an increased risk of having an allergic reaction to CT scan contrast dye. Kidney function: iodinated contrast agents can cause acute kidney injury, and patients should be evaluated for related risks before the surgery. Combination medication: drugs that may increase the risk of adverse reactions to iodinated contrast agents: neuroleptics and antidepressants, interleukin-2, beta-blockers, metformin. Situations where iodinated contrast agents should be used with caution: lung and heart diseases, catecholamine secreting tumours, pregnant and lactating women, myeloma and macroglobulinemia, myasthenia gravis, homocystinuria. Contraindication: uncured patients with hyperthyroidism. Dose limit for iodinated contrast agents On the premise of satisfying imaging/diagnosis, the minimum dose of iodinated contrast agents should be used. For low-risk patients, the total dose of types of contrast agents had better be controlled within 300 ~ 400 ml. Maximum dose of iodinated radiocontrast dye Method I *: 5 ml × body weight (kg)/serum creatinine (mg/dl) (not more than 300 ml) [refer to Cigarroa calculation formula] Method II: 3.7 times creatinine clearance [according to 2011 ACCF/AHAUA/NSTEMI Treatment Guidelines] For those with severe renal insufficiency, try to choose an imaging method that does not require iodine-based contrast agents or a non-imaging method that can provide sufficient diagnostic information. Avoid repeated use of the diagnostic dose of an iodinated contrast medium in a short period of time. If repeated use is indeed necessary, it is recommended that the interval between two MRI contrast agent applications be ≥14 days. Other precautions for the use of iodinated contrast agents Preheating: Before using an iodinated contrast agent, it is recommended to heat the contrast agent to 37 °C and place it in a thermostat. Hydration: It is recommended to hydrate patients within 6 to 12 hours before using an iodinated contrast agent to within 24 hours after use. Hydration method: Intra-arterial drug users are advised to receive intravenous rehydration and oral rehydration concomitantly, and oral rehydration is recommended for intravenous drug users. Stay and observation: After the injection of the iodine-based contrast media, the patient needs to stay and be observed for 30 minutes before leaving the examination room. Establish an emergency channel: Establish an emergency rapid support mechanism for the rescue of adverse reactions caused by iodinated contrast agents with the emergency room or other clinical related departments to ensure that after an adverse reaction occurs, clinicians can promptly rush to the rescue scene for rescue if necessary. Prevention and Management of Vascular Extravasation of Iodinated Contrast Agents Prevention Choose appropriate blood vessels for venipuncture and operate carefully; when using a high-pressure syringe, choose a puncture needle and catheter that match the injection flow rate; properly fix the puncture needle; communicate with the patient and get cooperation. Management Mild extravasation: Most injuries are minor and need no treatment. However, the patient should be instructed to keep observing. If the extravasation worsens, the patient should seek medical attention in time. For individuals with significant pain, common cold and hydropathic compresses can be given locally. Moderate and severe extravasation: ① raise the affected limb to promote blood return. ② Use 50% magnesium sulfate moisturizing cold compress in the beginning, and after 24-hours change to magnesium sulfate moisturizing hot compress, or use mucopolysaccharide ointment for topical application, or use 0.05% dexamethasone for local hydropathic compress. ③ Those with severe contrast agent extravasation should be given oral dexamethasone 5 mg/time tid for 3 consecutive days on the basis of topical drug use. ④ If necessary, consult a clinician for medication. Bei lu is a professional contrast media manufacturing companies, we provide ct contrast agent, mri contrast medium, contrast media products and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
For the purpose of medical imaging, a certain substance is introduced into the human body to change the image contrast of local tissues of the body. This introduced substance is Contrast Media, also known as Contrast Agents. As the first Contrast Media manufacturer in China, Beilu Pharma launched the domestic MRI Contrast Media Gadopentetate Dimeglumine Injection in 1992, which broke the foreign products monopoly and filled the domestic gap. As a well-known contrast media manufacturing companies, we could provide a broad portfolio stems Contrast Media across CT/X-ray and MRI modalities, and own the EU standard manufacturing facilities. Types of Contrast Media in Beilu Pharma Magnetic Resonance Imaging (MRI)Contrast Media is Gadolinium based contrast agent(GBCA). It is an intravenous injection, mainly used to enhance the image quality of internal organs, blood vessels and tissues during MRI examinations. GBCA contains gadolinium, which is connected to each other through carrier molecules to form a chelating agent. According to the different chemical structures, this contrast medium can be divided into two categories: linear and macrocyclic. The linear structure of the imaging agents is easier to release gadolinium ions, and the macrocyclic structure of the contrast material is more stable and less likely to release gadolinium ions. CT/X-RAY CONTRAST MEDIA Iodine-containing Contrast Media is one of the common imaging contrast agents used in radiology diagnosis and interventional therapy. It is also a basic diagnosis drug for cardiovascular imaging under X-ray. Currently, the iodinated Contrast Media used for cardiovascular system CT and digital subtraction angiography are all water-soluble organic iodinated contrast materials. Finished Types of Contrast Media S. No Product name Characteristic Specification 1 Gadopentetate Dimeglumine Injection Injectable solution 469mg/ml: 10ml, 12ml, 15ml, 20ml 2 Gadobutrol Injection Injectable solution 604.72mg/ml: 7.5ml 3 Ammonium iron citrate Oral granules 3g:0.6g(Fe 129mg) 6g:1.2g(Fe 258mg) 4 Iopamidol Injection Injectable solution 300mgI/ml: 50ml, 100ml 370mgI/ml: 50ml, 100ml 5 Iodixanol Injection Injectable solution 270mgI/ml: 100ml 320mgI/ml: 50ml, 100ml 6 Iohexol Injection Injectable solution 300mgI/ml: 20ml,50ml,75ml, 100ml 350mgI/ml: 20ml, 50ml, 100ml API & Intermediates Types of Contrast Media S. No Products CAS NO. Packings Classification 1 Iohexol 66108-95-0 25kg Contrast agent 2 Iodixanol 92339-11-2 25kg Contrast agent 3 Iopromide 73334-07-3 25kg Contrast agent 4 Iopamidol 60166-93-0 25kg Contrast agent 5 Ioversol 87771-40-2 25kg Contrast agent 6 Gadopentetate Monomeglumine 92923-57-4 25kg Contrast agent 7 Gadopentetate Dimeglumine 86050-77-3 25kg Contrast agent 8 Gadobutrol 138071-82-6 25kg Contrast agent 9 Calcobutrol 151878-23-8 2kg Excipients 10 Repaglinide 135062-02-1 10kg Anti-diabetic 11 Diethylenetriaminepentaacetic acid ( Pentaacetic Acid)(DTPA) 67-43-6 2.5kg Intermediates 12 Iopamidol Intermediate (order based) 5-Amino-2,4,6-triiodoisophthalic acid 35453-19-1 25kg Intermediates Comparison of physicochemical characteristics of commonly used Contrast Media Contrast media (Concentration:mg(I)/ml) Osmotic pressure (mOsm/kg water) Viscosity (cP,37℃) Iopamidol 300 616 4.7 Ioverol 300 651 5.5 Iohexol 300 672 6.3 Iobitridol 300 695 6 Iodixanol 320 290 11.8 The osmotic pressure of normal people is about 290-300mOsm/kg. Taking the viscosity of water as 1, when the temperature is 37℃, the relative viscosity of whole blood is 4-5, and the relative viscosity of plasma is 1.6- 2.4. Three factors of renal injury induced by Contrast Media: Osmotic pressure, Viscosity and the total iodine. Reduced iodine intake is a feasible strategy to avoid kidney damage. Beilu is a professional contrast media companies, we provide ct contrast agent, mri contrast medium, contrast media products and etc. Want to know more? Please contact us.
Minimum Order: 20 square metres
Phone:
Fax:
Email:
No.3 Shuiyuan West Road, Miyun District, Beijing,China
Beijing 101509
China
SOURCE: Import-Export Bulletin Board (https://www.imexbb.com/)
| Post an Offer to Sell |
| HomeOffers to BuyBusiness OpportunitiesCompany Profiles |
© 1996-2026 IMEXBB.com. All rights reserved.
|
|
|